Launch Your Pipeline to Market Faster,
Without Growing Your Team
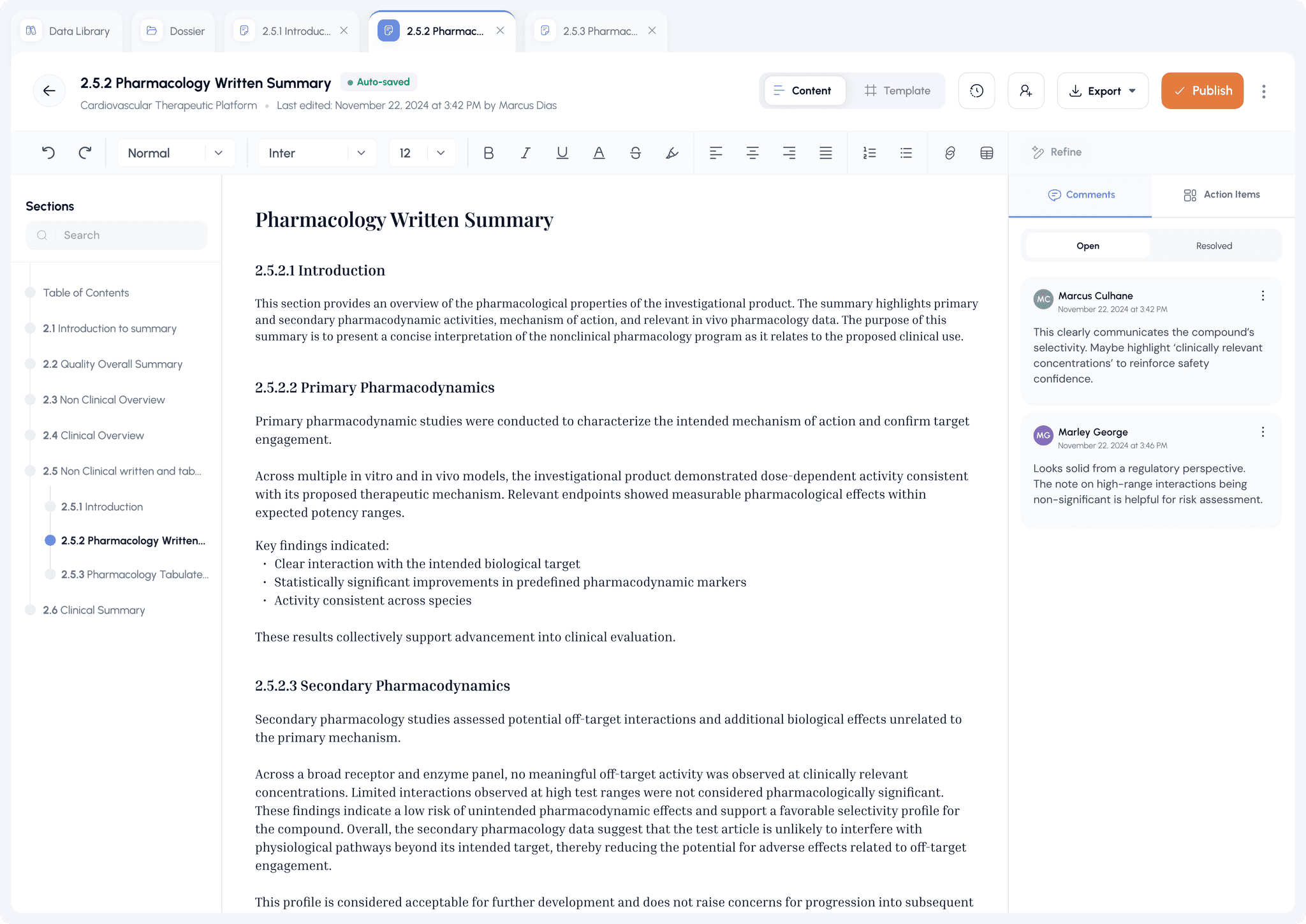
Launch Your Pipeline to Market Faster,
Without Growing Your Team
Zenopsys is the AI platform for authoring, reviewing, and submitting every document across drug development — clinical protocols, CSRs, and full eCTD dossiers (IND, ANDA, NDA, MAA). Built for pharma innovators, generics manufacturers, and regulatory consultants worldwide.
Book a Demo





Trusted by India's leading Life Sciences Companies
Benefits
Outcomes that Redefine Regulatory Efficiency


10× Faster Documentation Cycles
End-to-end, source data to submission-ready draft


First Drafts in Minutes, Not Weeks
Across every document type and use case


99.9% Manual Error Reduction
Significantly reduce errors with AI Review agents catching gaps pre-review
Platform Capabilities
Capabilities that Bring Structure to Regulatory Work
Dossier Authoring
With a single click, turn source data into structured, submission-ready technical documents in minutes.




AI Regulator
AI-powered review that thinks, checks, and guides teams to identify gaps — like a real FDA regulator.
Private “ChatGPT”
Chat with the largest guidance, webinar repository & internal databases. The only AI assistant a regulatory professional will ever need!



End-to-End Traceability
Maintain clear traceability across source data, document sections, and revisions so reviewers can quickly understand what changed, why, and where - accelerating review cycles.
Iterate Faster with AI
Create tables, lists, figures, and apply document styles with ease using our built-in “Refine with AI”. Eliminate the lengthy manual work and last-minute rework!




Built-In Collaboration
Collaborate in real time with shared access, comments, and version control. Keep teams aligned across drafting, review, and preparation stages without version confusion or email-based reviews.
Testimonials
Proven in Live Regulatory Submissions
Proven in Live
Regulatory Submissions

Dr. Jayanth Sridhar
Ex. CEO, Biologics, Dr. Reddy's
“As a CEO, if I can accelerate time to market by even 2 months that's a huge impact on my top-line. In an extremely competitive biosimilars and generics market, what Zenopsys has built will be a game changer.”

Anand Ekambaram
Ex. VP, Merck, BMS (USA)
“At Merck, it took us 3-4 months of back and forth reviews on all CMC, Clinical documents to identify any gaps or inconsistencies and get submission ready. It amazes me how Zenopsys has built an AI reviewer that eliminates all of that effort with real-time quality checks.”

Surya Namburi,
Director, iPharMed (UK Based Regulatory Consulting Firm)
"Zenopsys cut our Module 2.4, 2.5 authoring time from 4 weeks to under 2 days, and the output went into our submission with minimal edits. The traceability gave our QA team confidence we'd never had with manual drafts."

Dr. Jayanth Sridhar
Ex. CEO, Biologics, Dr. Reddy's
“As a CEO, if I can accelerate time to market by even 2 months that's a huge impact on my top-line. In an extremely competitive biosimilars and generics market, what Zenopsys has built will be a game changer.”

Anand Ekambaram
Ex. VP, Merck, BMS (USA)
“At Merck, it took us 3-4 months of back and forth reviews on all CMC, Clinical documents to identify any gaps or inconsistencies and get submission ready. It amazes me how Zenopsys has built an AI reviewer that eliminates all of that effort with real-time quality checks.”

Surya Namburi,
Director, iPharMed (UK Based Regulatory Consulting Firm)
"Zenopsys cut our Module 2.4, 2.5 authoring time from 4 weeks to under 2 days, and the output went into our submission with minimal edits. The traceability gave our QA team confidence we'd never had with manual drafts."

Dr. Jayanth Sridhar
Ex. CEO, Biologics, Dr. Reddy's
“As a CEO, if I can accelerate time to market by even 2 months that's a huge impact on my top-line. In an extremely competitive biosimilars and generics market, what Zenopsys has built will be a game changer.”

Anand Ekambaram
Ex. VP, Merck, BMS (USA)
“At Merck, it took us 3-4 months of back and forth reviews on all CMC, Clinical documents to identify any gaps or inconsistencies and get submission ready. It amazes me how Zenopsys has built an AI reviewer that eliminates all of that effort with real-time quality checks.”

Surya Namburi,
Director, iPharMed (UK Based Regulatory Consulting Firm)
"Zenopsys cut our Module 2.4, 2.5 authoring time from 4 weeks to under 2 days, and the output went into our submission with minimal edits. The traceability gave our QA team confidence we'd never had with manual drafts."

Dr. Jayanth Sridhar
Ex. CEO, Biologics, Dr. Reddy's
“As a CEO, if I can accelerate time to market by even 2 months that's a huge impact on my top-line. In an extremely competitive biosimilars and generics market, what Zenopsys has built will be a game changer.”

Anand Ekambaram
Ex. VP, Merck, BMS (USA)
“At Merck, it took us 3-4 months of back and forth reviews on all CMC, Clinical documents to identify any gaps or inconsistencies and get submission ready. It amazes me how Zenopsys has built an AI reviewer that eliminates all of that effort with real-time quality checks.”

Surya Namburi,
Director, iPharMed (UK Based Regulatory Consulting Firm)
"Zenopsys cut our Module 2.4, 2.5 authoring time from 4 weeks to under 2 days, and the output went into our submission with minimal edits. The traceability gave our QA team confidence we'd never had with manual drafts."

Dr. Jayanth Sridhar
Ex. CEO, Biologics, Dr. Reddy's
“As a CEO, if I can accelerate time to market by even 2 months that's a huge impact on my top-line. In an extremely competitive biosimilars and generics market, what Zenopsys has built will be a game changer.”

Anand Ekambaram
Ex. VP, Merck, BMS (USA)
“At Merck, it took us 3-4 months of back and forth reviews on all CMC, Clinical documents to identify any gaps or inconsistencies and get submission ready. It amazes me how Zenopsys has built an AI reviewer that eliminates all of that effort with real-time quality checks.”

Surya Namburi,
Director, iPharMed (UK Based Regulatory Consulting Firm)
"Zenopsys cut our Module 2.4, 2.5 authoring time from 4 weeks to under 2 days, and the output went into our submission with minimal edits. The traceability gave our QA team confidence we'd never had with manual drafts."

Dr. Jayanth Sridhar
Ex. CEO, Biologics, Dr. Reddy's
“As a CEO, if I can accelerate time to market by even 2 months that's a huge impact on my top-line. In an extremely competitive biosimilars and generics market, what Zenopsys has built will be a game changer.”

Anand Ekambaram
Ex. VP, Merck, BMS (USA)
“At Merck, it took us 3-4 months of back and forth reviews on all CMC, Clinical documents to identify any gaps or inconsistencies and get submission ready. It amazes me how Zenopsys has built an AI reviewer that eliminates all of that effort with real-time quality checks.”

Surya Namburi,
Director, iPharMed (UK Based Regulatory Consulting Firm)
"Zenopsys cut our Module 2.4, 2.5 authoring time from 4 weeks to under 2 days, and the output went into our submission with minimal edits. The traceability gave our QA team confidence we'd never had with manual drafts."

Dr. Jayanth Sridhar
Ex. CEO, Biologics, Dr. Reddy's
“As a CEO, if I can accelerate time to market by even 2 months that's a huge impact on my top-line. In an extremely competitive biosimilars and generics market, what Zenopsys has built will be a game changer.”

Anand Ekambaram
Ex. VP, Merck, BMS (USA)
“At Merck, it took us 3-4 months of back and forth reviews on all CMC, Clinical documents to identify any gaps or inconsistencies and get submission ready. It amazes me how Zenopsys has built an AI reviewer that eliminates all of that effort with real-time quality checks.”

Surya Namburi,
Director, iPharMed (UK Based Regulatory Consulting Firm)
"Zenopsys cut our Module 2.4, 2.5 authoring time from 4 weeks to under 2 days, and the output went into our submission with minimal edits. The traceability gave our QA team confidence we'd never had with manual drafts."

Dr. Jayanth Sridhar
Ex. CEO, Biologics, Dr. Reddy's
“As a CEO, if I can accelerate time to market by even 2 months that's a huge impact on my top-line. In an extremely competitive biosimilars and generics market, what Zenopsys has built will be a game changer.”

Anand Ekambaram
Ex. VP, Merck, BMS (USA)
“At Merck, it took us 3-4 months of back and forth reviews on all CMC, Clinical documents to identify any gaps or inconsistencies and get submission ready. It amazes me how Zenopsys has built an AI reviewer that eliminates all of that effort with real-time quality checks.”

Surya Namburi,
Director, iPharMed (UK Based Regulatory Consulting Firm)
"Zenopsys cut our Module 2.4, 2.5 authoring time from 4 weeks to under 2 days, and the output went into our submission with minimal edits. The traceability gave our QA team confidence we'd never had with manual drafts."
Enterprise Control & Security
Enterprise Control & Security
Designed for Enterprise Trust and Control
Designed for Enterprise Trust and Control
Zero Data Storage and Zero Training Agreement with LLM Providers
Your regulatory data is not stored or used to train external LLM providers. All processing is designed to ensure sensitive content remains under your control, supporting strict confidentiality and data-privacy requirements.
Zero Data Storage and Zero Training Agreement with LLM Providers
Your regulatory data is not stored or used to train external LLM providers. All processing is designed to ensure sensitive content remains under your control, supporting strict confidentiality and data-privacy requirements.
Enterprise-Grade Security
Built to align with recognized enterprise security and privacy standards, including ISO, SOC 2, HIPAA, and GDPR—supporting deployment in regulated and high-trust environments.
Enterprise-Grade Security
Built to align with recognized enterprise security and privacy standards, including ISO, SOC 2, HIPAA, and GDPR—supporting deployment in regulated and high-trust environments.
IP Whitelisting and Network Access Control
Deploy the platform with IP address whitelisting to restrict access at the network level. This ensures only approved enterprise networks can access the system, adding an extra layer of security.
IP Whitelisting and Network Access Control
Deploy the platform with IP address whitelisting to restrict access at the network level. This ensures only approved enterprise networks can access the system, adding an extra layer of security.
Industries we support
Supporting Regulatory Work across Life Sciences

Biotech

Pharma

CRDMOs

Consultants
Investors & advisors
Backed by Experienced Operators and Domain Experts

Anand Ekambaram
Ex-VP, Merck & BMS (USA)
Advisor

Dr. Jayanth Sridhar
Ex-CEO, Dr. Reddy’s Biologics
Investor & Advisor

Venkat Kamalakar
CEO, Marksans Pharma, EU
Investor & Advisor

Dr. Mahesh Bhalgat
CEO, Veeda Life Sciences
Advisor

Srinivas Rao Aluri
Ex. MD, Morgan Stanley PE
Advisor

Funded by leading venture capital firms


Coverage
Built for Global Submissions
Zenopsys supports submissions across all major regulatory agencies and markets






Ready to Transform Your Regulatory Work
Book a Demo
Testimonials
Proven in Live
Regulatory Submissions

Dr. Jayanth Sridhar
Ex. CEO, Biologics, Dr. Reddy's
“As a CEO, if I can accelerate time to market by even 2 months that's a huge impact on my top-line. In an extremely competitive biosimilars and generics market, what Zenopsys has built will be a game changer.”

Anand Ekambaram
Ex. VP, Merck, BMS (USA)
“At Merck, it took us 3-4 months of back and forth reviews on all CMC, Clinical documents to identify any gaps or inconsistencies and get submission ready. It amazes me how Zenopsys has built an AI reviewer that eliminates all of that effort with real-time quality checks.”

Surya Namburi,
Director, iPharMed (UK Based Regulatory Consulting Firm)
"Zenopsys cut our Module 2.4, 2.5 authoring time from 4 weeks to under 2 days, and the output went into our submission with minimal edits. The traceability gave our QA team confidence we'd never had with manual drafts."

Dr. Jayanth Sridhar
Ex. CEO, Biologics, Dr. Reddy's
“As a CEO, if I can accelerate time to market by even 2 months that's a huge impact on my top-line. In an extremely competitive biosimilars and generics market, what Zenopsys has built will be a game changer.”

Anand Ekambaram
Ex. VP, Merck, BMS (USA)
“At Merck, it took us 3-4 months of back and forth reviews on all CMC, Clinical documents to identify any gaps or inconsistencies and get submission ready. It amazes me how Zenopsys has built an AI reviewer that eliminates all of that effort with real-time quality checks.”

Surya Namburi,
Director, iPharMed (UK Based Regulatory Consulting Firm)
"Zenopsys cut our Module 2.4, 2.5 authoring time from 4 weeks to under 2 days, and the output went into our submission with minimal edits. The traceability gave our QA team confidence we'd never had with manual drafts."

Dr. Jayanth Sridhar
Ex. CEO, Biologics, Dr. Reddy's
“As a CEO, if I can accelerate time to market by even 2 months that's a huge impact on my top-line. In an extremely competitive biosimilars and generics market, what Zenopsys has built will be a game changer.”

Anand Ekambaram
Ex. VP, Merck, BMS (USA)
“At Merck, it took us 3-4 months of back and forth reviews on all CMC, Clinical documents to identify any gaps or inconsistencies and get submission ready. It amazes me how Zenopsys has built an AI reviewer that eliminates all of that effort with real-time quality checks.”

Surya Namburi,
Director, iPharMed (UK Based Regulatory Consulting Firm)
"Zenopsys cut our Module 2.4, 2.5 authoring time from 4 weeks to under 2 days, and the output went into our submission with minimal edits. The traceability gave our QA team confidence we'd never had with manual drafts."

Dr. Jayanth Sridhar
Ex. CEO, Biologics, Dr. Reddy's
“As a CEO, if I can accelerate time to market by even 2 months that's a huge impact on my top-line. In an extremely competitive biosimilars and generics market, what Zenopsys has built will be a game changer.”

Anand Ekambaram
Ex. VP, Merck, BMS (USA)
“At Merck, it took us 3-4 months of back and forth reviews on all CMC, Clinical documents to identify any gaps or inconsistencies and get submission ready. It amazes me how Zenopsys has built an AI reviewer that eliminates all of that effort with real-time quality checks.”

Surya Namburi,
Director, iPharMed (UK Based Regulatory Consulting Firm)
"Zenopsys cut our Module 2.4, 2.5 authoring time from 4 weeks to under 2 days, and the output went into our submission with minimal edits. The traceability gave our QA team confidence we'd never had with manual drafts."
Coverage
Built for Global Submissions
Zenopsys supports submissions across all major regulatory agencies & markets






Trusted by India's leading Life Sciences Companies
Benefits
Outcomes that Redefine Regulatory Efficiency


10× Faster Documentation Cycles
End-to-end, source data to submission-ready draft


First Drafts in Minutes, Not Weeks
Across every document type and use case


99.9% Manual Error Reduction
Significantly reduce errors with AI Review agents catching gaps pre-review
Platform Capabilities
Capabilities that Bring Structure to Regulatory Work
Dossier Authoring
With a single click, turn source data into structured, submission-ready technical documents in minutes.




AI Regulator
AI-powered review that thinks, checks, and guides teams to identify gaps — like a real FDA regulator.




Private “ChatGPT”
Chat with the largest guidance, webinar repository & internal databases. The only AI assistant a regulatory professional will ever need!




End-to-End Traceability
Maintain clear traceability across source data, document sections, and revisions so reviewers can quickly understand what changed, why, and where - accelerating review cycles.




Iterate Faster with AI
Create tables, lists, figures, and apply document styles with ease using our built-in “Refine with AI”. Eliminate the lengthy manual work and last-minute rework!




Built-In Collaboration
Collaborate in real time with shared access, comments, and version control. Keep teams aligned across drafting, review, and preparation stages without version confusion or email-based reviews.




Industries we support
Supporting Regulatory Work across Life Sciences

Biotech

Pharma

CRDMOs

Consultants
Investors & advisors
Backed by Experienced Operators and Domain Experts

Anand Ekambaram
Ex-VP, Merck & BMS (USA)
Advisor

Dr. Jayanth Sridhar
Ex-CEO, Dr. Reddy’s Biologics
Investor & Advisor

Venkat Kamalakar
CEO, Marksans Pharma, EU
Investor & Advisor

Dr. Mahesh Bhalgat
CEO, Veeda Life Sciences
Advisor

Srinivas Rao Aluri
Ex. MD, Morgan Stanley PE
Advisor

Funded by leading venture capital firms




Coverage
Built for Global Submissions
Zenopsys supports submissions across all major regulatory agencies & markets






Ready to transform your regulatory work
Book a Demo
Platform Capabilities
Capabilities that Bring Structure to Regulatory Work
Dossier Authoring
With a single click, turn source data into structured, submission-ready technical documents in minutes.








AI Regulator
AI-powered review that thinks, checks, and guides teams to identify gaps — like a real FDA regulator.
Private “ChatGPT”
Chat with the largest guidance, webinar repository & internal databases. The only AI assistant a regulatory professional will ever need!








End-to-End Traceability
Maintain clear traceability across source data, document sections, and revisions so reviewers can quickly understand what changed, why, and where - accelerating review cycles.
Iterate Faster with AI
Create tables, lists, figures, and apply document styles with ease using our built-in “Refine with AI”. Eliminate the lengthy manual work and last-minute rework!








Built-In Collaboration
Collaborate in real time with shared access, comments, and version control. Keep teams aligned across drafting, review, and preparation stages without version confusion or email-based reviews.
Investors & advisors
Backed by Experienced Operators and Domain Experts

Anand Ekambaram
Ex-VP, Merck & BMS (USA)
Advisor

Dr. Jayanth Sridhar
Ex-CEO, Dr. Reddy’s Biologics
Investor & Advisor

Venkat Kamalakar
CEO, Marksans Pharma, EU
Investor & Advisor

Dr. Mahesh Bhalgat
CEO, Veeda Life Sciences
Advisor

Srinivas Rao Aluri
Ex. MD, Morgan Stanley PE
Advisor

Funded by leading venture capital firms




Ready to transform your regulatory work
Book a Demo











